
By Mike Darwin
Preparing the Patient for Shipment
Closed chest cardiopulmonary support (CPS) will generally be discontinued either at the start of in-field cardiopulmonary bypass, or when the patient’s frontal sinus or tympanic temperature reaches ~ 15ºC. Due to hypothermia-induced blood sludging, cold agglutination and red blood cell (RBC) aggregation, continuation of CPS below 15ºC is currently deemed to be ineffective and possibly counterproductive.
Many patients will be remote from the facility where cryoprotective perfusion will be carried out and will be transported by common carrier or private carrier over considerable geographical distances. In some cases it will be possible to move the patient using a specialized transport vehicle with on-going extracorporeal support. In other cases the distances will be sufficiently great that the only realistic option is iced-shipment in the absence of perfusion. It is often necessary to use a commercial air freight service to move the patient from one area of the United States to another (or from one country to another).
Preparation Following Extracorporeal Support
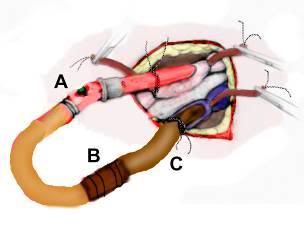 Figure 1: The arterial (A) and venous lines (C) should be cross-connected using a 3/8 “x 1/2″ connector (B) with great care being taken to avoid introducing air into the tubing or connector during this operation. A syringe may be used to dribble perfusate or normal saline into the opening between the two lines/connector as they are approximated and connected. The edges of the wound are then approximated around the cannulae and stapled closed.
Figure 1: The arterial (A) and venous lines (C) should be cross-connected using a 3/8 “x 1/2″ connector (B) with great care being taken to avoid introducing air into the tubing or connector during this operation. A syringe may be used to dribble perfusate or normal saline into the opening between the two lines/connector as they are approximated and connected. The edges of the wound are then approximated around the cannulae and stapled closed.
If extracorporeal support and/or blood washout are to follow CPS then a considerable amount of preparation of the patient for shipment will be necessary at the conclusion of extracorporeal support. Once blood washout and cooling to 2-4ºC are achieved, the patient will need to be disconnected from the extracorporeal circuit. This should be done by cutting or “breaking” the arterial and venous lines aseptically and cross-connecting them (avoiding any introduction of air) with a 3/8″-1/2″ sterile connector (Figure 1). The edges of the groin wound(s) are then approximated with the cannulae still in place, the skin at the operative site is toweled dry with a sterile muslin towel or gauze 4x4s, and a waterproof 3-M adhesive drape is placed over the wound and cannulae to prevent water from entering the wound and to stabilize the cannulae against dislodgment.
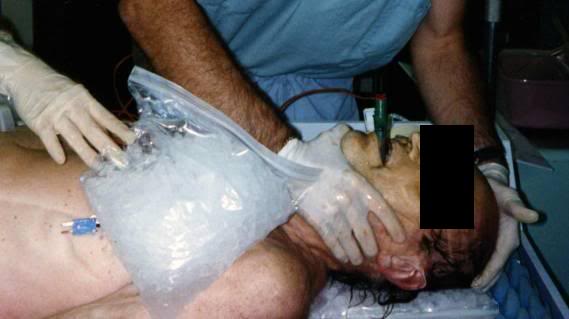 Figure 2: Combitube in position and capped after arrival of a patient from remote stabilization and blood washout.
Figure 2: Combitube in position and capped after arrival of a patient from remote stabilization and blood washout.
All other lines and catheters should then be capped or clamped using disposable plastic occluding clamps, Kelley clamps, or stainless steel tube occluding forceps, starting with those at the head and working down.
1) If present, the endotracheal tube is plugged or clamped. If either the DEGTA or Combitube are present, the mask is disconnected from the gastric tube/obturator; the gastric tube/obturator is clamped. If the Combitube has been used it should be left in position and plugged, not clamped. If an ET tube or DEGTA were used, and the mouth is packed with 1-2 4×4 gauze (to guard against water entering the airway) with a visible tail of each gauze square left outside the mouth to facilitate easy removal. Do not remove the DEGTA gastric tube/obturator, or let the balloon down, as this may allow seepage of gastric contents into the lungs.
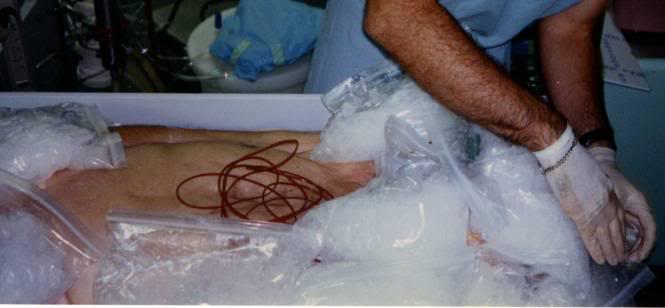 Figure 3: Probe leads on thorax following shipment of patient. The twist ties have just been removed and probe leads are ready for connection to the temperature monitoring equipment in the cryoprotective perfusion facility.
Figure 3: Probe leads on thorax following shipment of patient. The twist ties have just been removed and probe leads are ready for connection to the temperature monitoring equipment in the cryoprotective perfusion facility.
2) Disconnect all temperature probes from the monitor(s) and leave them in place. Probe leads are then coiled up neatly and secured in coils with twist ties. Coiled probe wires are placed on the ventral surface of the body where they may be secured with tape, if desired, to avoid being dislodged during movement of the patient from the PIB to the shipping container. If an auto-logging recording thermometer is being used to monitor the patient while in transit, it should be connected to the probe in closest proximity to the brain (nasal, pharyngeal, etc.). If a DuaLogR or similar recording thermometer is to be used ensure that it is in its watertight case and is placed on the patient’s thorax, well above the probable level of any melt water that may accumulate during shipping.
3) Any IV or central venous lines should be capped using aseptic technique. If caps are not available, the IV set-up must be clamped off and the entire IV set including bag or bottle left connected to the patient (and thus sterile).
4) The Foley catheter is plugged or clamped. It is not necessary to maintain sterility of the distal end of the catheter.
5) Rectal and peritoneal lavage tubes are left in place and the tubing plugged or clamped. The rectal tube line should be wrapped in several gauze 4x4s to prevent the spread of any feces contaminated drainage.
The patient’s eyes are closed with tape and all visible blood and secretions on the body are cleaned up while the patient still in the PIB. Be certain that blood and other laboratory samples are attached to the right forearm with tape!
The patient’s hands are positioned at his or her side and the legs are fully extended and placed in close approximation to each other. The PIB is then drained of water as completely as possible using the SCCD, or by bailing the water out manually.
Before the patient is removed from the PIB, the air shipment container should be prepared by placing a layer of Zip-Loc bags filled with flaked or small cubed ice over the bottom of the inner container of the shipper. If a heavy-duty body bag is available it should be used to hold the ice bags that will go under the patient. Enough additional ice bags should be prepared in advance to allow for complete and thorough packing of the patient in ice immediately upon transfer to the shipper. Generally, 50 kg (110 lbs) of ice are required to safely refrigerate a patient (who has been pre-cooled to near 5ºC) for 24 to 48 hours in a well insulated[1] container. The nomogram (Figure 4) will give approximate ice melt values per unit of time for a pre-cooled patient in a metal shipping container insulated with either 2” of expanded polystyrene (EPS) or ½” Rmax R-Matte Plus-3 foil-clad polyurethane foam board, with a stated R value of 3.2.
More ice will be needed if any of the following conditions pertain:
- The ambient temperature is significantly above 24 ºC.
- The patient weighs significantly more than 80 kg (176 lb).
- The patient’s body temperature at the time of packing for shipping is greater than 5ºC.
- Shipping time is anticipated to longer than 36 hours.
Immediately prior to transferring the patient to the shipment container, excess ice should be removed from the PIB and/or the patient should be elevated in the PIB and the ice should be moved under the patient to raise the patient above any remaining liquid on a bed of ice. The patient is then wiped down with towels while the dorsal surface of the body is allowed to drip free of excess fluid on the bed of ice. A c\temp disposable electronic temperature logger (http://www.temperature-data-logger.com/c_temp.html) should be placed over the patient’s right temple using the accompanying Velcro headband. The c\temp should be set to record temperatures at 58 second intervals (e.g., 5 day setting).The purpose of the temperature data logger is provide quality assurance during patient air shipment that there has been no warming and to provide a record of the patient’s transport temperature for later clinical and scientific evaluation (most patients will still be well above ~ 1ºC when they are placed in the air shipper. The c\temp, along with a representative data stream, is shown in Figure 4, below.
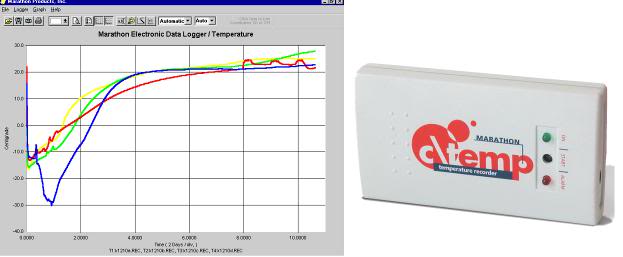
Figure 4: The c\temp dlt temperature logger operates in therange of -81°C to +30°C and may be used for both water ice and dry ice shipment of cryonics patients. The device has a lithium battery with a 2-year shelf life and can acquire data at intervals as brief as 50 seconds for up to 5 days. It measures 9(L) x 5(W) x 1.6(H) cm and 58 grams. Data is downloaded to a PC using proprietary software.
Once the patient is cleaned up and relatively dry, place the patient’s head in a plastic bag (to protect the airway and ophthalmic and nasopharyngeal mucosa from exposure to water) and lightly secure the bag around the patient’s beck with tape. The patient is then moved to the shipment container. If a body bag is being used as a liner to the inner box of the shipment container, it should be filled with Zip-Loc bags containing crushed or small cubed ice.
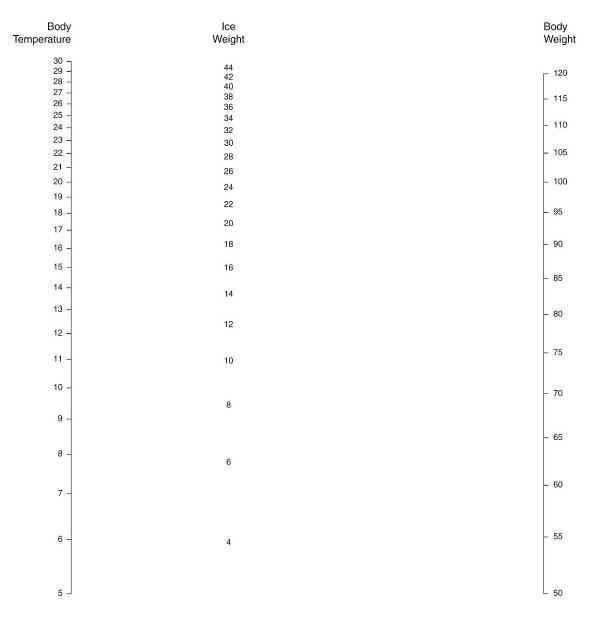 Figure 5: Nomogram for determining the minimum safe amount of ice required for patient air transport. To determine the amount of ice that will be needed based on the patient’s body weight and temperature place a straight edge between the body weight scale on the right and the patient body weight scale on the left. The quantity of ice that will be consumed (melted) kilograms (kg) is shown on the center scale. Body weight is in kg and temperature is in ºC. Ambient temperature is assumed to be ~24.ºC. Reproduced from: “Ice Melting Time in a Patient Shipment Container by Charles Platt, Aschwin de Wolf, and Jay Wasserlauf; Suspended Animation, Inc, April 2005.”[4]
Figure 5: Nomogram for determining the minimum safe amount of ice required for patient air transport. To determine the amount of ice that will be needed based on the patient’s body weight and temperature place a straight edge between the body weight scale on the right and the patient body weight scale on the left. The quantity of ice that will be consumed (melted) kilograms (kg) is shown on the center scale. Body weight is in kg and temperature is in ºC. Ambient temperature is assumed to be ~24.ºC. Reproduced from: “Ice Melting Time in a Patient Shipment Container by Charles Platt, Aschwin de Wolf, and Jay Wasserlauf; Suspended Animation, Inc, April 2005.”[4]
Note: It is very important that the patient’s abdomen and thorax not be heavily loaded with ice as the ice will exert a large hydrostatic force and will displace perfusate (or blood) from the heart and great vessels into the peripheral tissues. This is especially important in the case of patients who are being shipped without the benefit of blood washout, as loading of the chest and abdomen will force blood into the capillaries where ultrafiltration will occur through the capillary membranes. This results in hemoconcentration and plugging of the capillaries with high viscosity sludge of formed elements of the blood. Additionally, in patients where clotting has occurred there is the danger of forcing clots from the central circulation both retrograde and anteretrograde into the arterioles and venules where they may be impossible to dislodge.
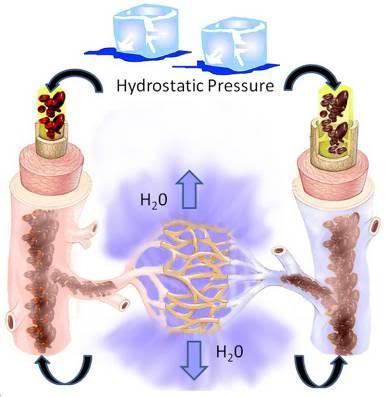 Figure 6: Loading the patient’s abdomen and thorax with ice creates hydrostatic pressure in the vasculature. This pressure will result in filtration of water from the blood via the microcirculation into the interstitial space. The result will be massive hemoconcentration of blood components and plasma proteins resulting in a viscous sludge that will be difficult or impossible to displace during cryoprotective perfusion.
Figure 6: Loading the patient’s abdomen and thorax with ice creates hydrostatic pressure in the vasculature. This pressure will result in filtration of water from the blood via the microcirculation into the interstitial space. The result will be massive hemoconcentration of blood components and plasma proteins resulting in a viscous sludge that will be difficult or impossible to displace during cryoprotective perfusion.
Do not overload the top (ventral surface) of the patient with ice; keep the ice bags on the abdomen and chest to no more than 1″ of ice thickness. Do not place large trash bags or other large plastic bags loaded with ice on top of the patient (See Figure 7)!
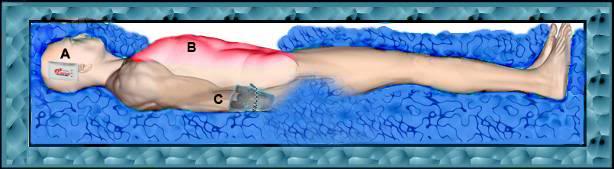
Figure 7: Do not load ice atop the patient’s thorax or abdomen (B). Ensure that the c\temp continuous temperature logger is placed next to the patient’s head (A) and that the patient’s laboratory and effluent effluent samples are affixed to his right arm (C).
Special attention should be given to completely packing the patient’s head in ice. Upon completion of ice packing, the body bag is closed (if it is being used) and the inner container is closed and sealed. The outer container is then closed and prepared for shipment.
Ideal Common Carrier Shipment Container
 Figure 8: Optimal purpose-built shipping container constructed of welded steel frame and heavy-duty plywood. Note the large number of handles and the special checkerboard marking.
Figure 8: Optimal purpose-built shipping container constructed of welded steel frame and heavy-duty plywood. Note the large number of handles and the special checkerboard marking.
Common carrier transport of the patient requires uncommon preparation and attention to detail if it is to be carried out safely. On two occasions in the past patients have suffered warming or have experienced delays of many hours in transportation as a result of leaking or inadequately insulated shipping containers.[1],[2] Whenever possible a purpose-built shipping container should be used for common carrier transport. A corollary of this recommendation is that a purpose built container should be both kept on hand, and be immediately ready for transport to the site where it will be used. Shipping containers should always be deployed with the Remote Standby Kit (RSK) when a Standby commences for a clearly terminal patient.
 Figure 9: Tamper evident seal placed on patient air shipper after inspection by air freight/TSA security officials prior to shipping. If the lid of the shipper is opened, or an attempt is made to remove the seal, it will be immediately evident.
Figure 9: Tamper evident seal placed on patient air shipper after inspection by air freight/TSA security officials prior to shipping. If the lid of the shipper is opened, or an attempt is made to remove the seal, it will be immediately evident.
- It is large enough to accommodate a large adult male (198 cm 110 kg) and still have an adequate volume for 100 kg of ice.
- Exterior walls are constructed from 3/4″ marine plywood with a welded steel frame making the shipper extremely durable and unlikely to come apart if it is dropped, or to be penetrated by forklift tines during handling.
- The unit has multiple handles on all sides to facilitate easy lifting by up to 10 people.
- The exterior lid requires a tool (a heavy-duty Phillips screwdriver) to be opened to discourage casual inspection of the contents, and can be tamper-evident sealed to detect unauthorized entry (Figure 9).
- The inner container is fabricated from welded, high-impact polypropylene which is fluid-tight, impact resistant, chemically impervious, and can be hermetically sealed. Polypropylene containers of this type also allows the unit to be used for dry ice shipment of patients should straight freezing be necessary.
- The inner container can be sealed in the field using a self-releasing silicone gasket forming compound which insures that no leakage of refrigerant or body fluids from around the patient can occur.
- The exterior of the container is marked both by its color (safety orange) and by a black and white checkerboard pattern which allows for easy identification during shipment or if it is delayed or accidentally misrouted. The checkerboard material is vinyl adhesive contact covering available from Kittrich, Corp., (order # 9P23 Con-Tact Brand Covering): http://www.amazon.com/Kittrich-Corp-Con-Tact-Brand-Covering/dp/B001ARHJ24.
 Figure 10: Thermometer and alarm assembly at the head-end of the shipment container. The thermometer is a battery-powered unit that is protected by a spring-loaded steel door. The thermometer can be set to sound an audible alarm and stores high and low temperatures in memory. An exterior 110 VAC electrical outlet cover used to protect the remote-sensing battery operated thermometer purchased from a general merchandise store (Wal-Mart).
Figure 10: Thermometer and alarm assembly at the head-end of the shipment container. The thermometer is a battery-powered unit that is protected by a spring-loaded steel door. The thermometer can be set to sound an audible alarm and stores high and low temperatures in memory. An exterior 110 VAC electrical outlet cover used to protect the remote-sensing battery operated thermometer purchased from a general merchandise store (Wal-Mart).
 Figure 11: Left: The lid of the shipping container fits flush onto the routed wood of the container box. The insulation for the top of the container is not attached to the lid, thus decreasing wear and tear on the insulation.
Figure 11: Left: The lid of the shipping container fits flush onto the routed wood of the container box. The insulation for the top of the container is not attached to the lid, thus decreasing wear and tear on the insulation.
As shown in Figure 10 the container has a battery operated LCD thermometer with alarm and high-low memory capability built into it which is armored against injury. This allows for monitoring of the patient’s temperature during shipment and immediately upon delivery without opening the container. These thermometers operate on a single AA alkaline cell for over a year.
The exterior of the container has a smooth profile with no latches or hinges for anything to catch on. The steel frame is completely smooth, and flush with the walls of the box and has no lip which can hang up on the conveyor belts or become entangled with adjacent freight causing the container to be thrown off the belt, or the frame to bend or deform (both of which happened with previous designs).
 Figure 12: Inner top insulating panel of expanded polystyrene foam is lifted to expose the inner liquid-tight box of welded polypropylene.
Figure 12: Inner top insulating panel of expanded polystyrene foam is lifted to expose the inner liquid-tight box of welded polypropylene.
 Figure 13: The lid of the inner container has been removed. Note the lip on the lid which allows creation of a non-adhering silicone gasket in the field using Blue RTV Form-A-Gasket, or similar material.
Figure 13: The lid of the inner container has been removed. Note the lip on the lid which allows creation of a non-adhering silicone gasket in the field using Blue RTV Form-A-Gasket, or similar material.
 Figure 14: Inner container showing thermometer assembly, sealing lip, spring-loaded catches at 6″ intervals, and boxes of Zip-Loc bags for ice packing inside.
Figure 14: Inner container showing thermometer assembly, sealing lip, spring-loaded catches at 6″ intervals, and boxes of Zip-Loc bags for ice packing inside.
On-Site Fabrication of a Shipment Container
All too often it will not be possible to have a purpose-built container available when needed. The next best alternative is to construct a quality shipper on site if time permits. The following sequence of photographs shows the key elements of such a field-built container. This container has been assembled from three elements which are available from most mortuaries: a sealer casket, a sheet metal air shipping box, and a standard airline casket air shipment tray. The other items necessary for putting such a container together (foil tape, foam or fiberglass building insulation, and a remote sensing (indoor/outdoor) thermometer, etc.) can be obtained from Radio Shack, Wal-Mart or any general purpose hardware or home supply outlet.
 Figure 15: An air tray containing a standard metal sealer casket. The air tray consists of a sturdy wooden base with straps to accept a heavy cardboard cover for the casket.
Figure 15: An air tray containing a standard metal sealer casket. The air tray consists of a sturdy wooden base with straps to accept a heavy cardboard cover for the casket.
In the past, very reliable water-tight and sealable metal air shipping containers (for routine transfer of human remains between mortuaries) were widely stocked by most mortuaries. Most of these containers were manufactured by Fayette Lock and Tool of Connersville, Indiana (the very best are re-usable shipment cases manufactured by Ziegler Metal Products, and are used almost exclusively by the military, and rarely if ever, by the civilian sector). In the mid-1980′s Fayette Lock and Tool ceased operation and the standard for air shippers dropped dramatically. Today, the typical mortuary air shipment box is crudely made of galvanized metal which is poorly soldered or tack-welded at the joins. Such shippers are not water tight and will leak profusely if filled with ice bags with or without a body bag as a liner.
NOTE: The importance of having a water tight, leak-proof shipment container cannot be overstated. Virtually all common carriers will reject a leaking container, will refuse to ship it if it has been accepted, and will pull it from shipment and notify local health authorities if it begins to leak during shipment.
Sealing the shipment container against leakage thus becomes of paramount importance. The first step in achieving this is to caulk all the joints of the inner mortuary air shipper with silicone caulk (bathtub or exterior silicone caulk is acceptable) after preparing the surfaces to be caulked by wiping with rubbing alcohol (70% isopropanol in water), acetone (nail polish remover) or other degreasing agents to insure good adhesion of the sealant.
A metal sealer casket is then stripped of its decorative lining and prepared to receive insulation. Foam board (urethane, expanded polystyrene, or other foam board insulation) or fiberglass wool insulation is laid down on the bottom of the sealer casket. The mortuary shipping box is placed atop the insulation and additional foam board is cut to insulate the sides and ends of the box by filling the annulus between the sealer casket and the mortuary shipper.
The lid of the sealer casket is filled with fiberglass wool and/or foam board insulation. The insulating material can be protected against water and improved in efficiency by using foil backed adhesive tape of the kind used in home gutter repairs to cover the foam board or glass wool.
 Figure 16: Sealer casket on an air tray under the cardboard cover.
Figure 16: Sealer casket on an air tray under the cardboard cover.

Figure 17: Sealer casket with insulation in the lid, additional foam board insulation between the casket lid and mortuary shipping box, and foam board and fiberglass insulation between the inner and outer containers (casket and mortuary shipper).
In a situation where there is no time to obtain insulation and adhesives it will be necessary to rely on the integrity of the sealer casket to stop water leaks. Blankets, paper towels, wadded up newspaper, bubble wrap, Styrofoam peanuts, or any of a variety of materials may be used for improvised insulation. Whenever possible use absorbent material on the bottom of the sealer casket to absorb any leakage or sweating from the inner container. A multilayered defense against leakage is the best strategy.
 Figure 18: Interior of a sealer casket which has been outfitted as an in-field air shipper. A galvanized metal liner has been silicone caulked and a body bag and Zip-Loc bags are present inside to be used when the need arises.
Figure 18: Interior of a sealer casket which has been outfitted as an in-field air shipper. A galvanized metal liner has been silicone caulked and a body bag and Zip-Loc bags are present inside to be used when the need arises.
It is important to be aware that actual leakage is not the only potential problem which may cause interruption of shipment and quarantine of a patient in transit. Even if a water-tight container is used, sweating due to water condensation on cold metal may be mistaken for leakage. Thus, it is very important to have some insulation between the container holding the patient and refrigerant, and the outside of the shipper.
 Figure 19: Thermometer/alarm attached to the outside of a sealer casket with self-adhesive Velcro strips. Velcro and thermometer/alarms such as the one above can be purchased at Radio Shack, Wal-Mart or other general merchandise stores.
Figure 19: Thermometer/alarm attached to the outside of a sealer casket with self-adhesive Velcro strips. Velcro and thermometer/alarms such as the one above can be purchased at Radio Shack, Wal-Mart or other general merchandise stores.
Sealer Casket Operation
Sealer caskets are made by a number of manufacturers; however, the largest of these is the Batesville Casket Company in Batesville, Indiana. The Batesville sealer is the prototypical sealer casket. A low cost sealer consists of a metal box of 20 gauge steel with a lid which has a rubber gasket and a compression closing mechanism. The metal shell typically has bar-type handles which are attached with penetrating screws (which will leak if the casket is filled to handle height!). Welds are frequently shoddy, are typically backed up with pitch coating, and will leak (usually only slightly) if the container is even partially filled with liquid. The interior consists of fabric, and excelsior or polyester stuffing which is lightly attached to a tack strip of wood, or glued to the metal exterior.
This lining may be ripped away and used (if necessary) as absorbent material for the bottom of shipper. Most sealer caskets will have at least a rudimentary foam mattress and spring and wire tray or bed assembly to receive the body and adjust its height up or down in the casket. This frame should be removed if the casket is to be thoroughly insulated. Low-end sealers cost about $800 US. Mortuary air shippers range in cost from $150 to $175, US.
 Figure 20: Gasketed sealing screw-port which covers the lid-locking mechanism of a typical sealer casket.
Figure 20: Gasketed sealing screw-port which covers the lid-locking mechanism of a typical sealer casket.
At the foot end of the sealer on the lip of the box just under the lid is a port capped by a gasketed screw-plug (see Figure 20). The plug is removed to reveal a recessed female hex head bolt which a hex-wrench can be inserted into to lock-down the lid. Typically sealers will come with a hex-handled closure tool (often decorative) called a “casket key.” This tool should be carefully stowed with the casket after it is closed so that the casket can be rapidly and safely opened when it arrives. The casket key hex wrench is of a large non-metric size and is not commonly found in home supply or nonprofessional hardware stores. If the casket key is lost opening the sealer can be very difficult and may require the use of power tools. Additionally, a few manufacturers use different hex-head sizes and, more rarely, unusual key shapes.
It is important to lock the casket with the key prior to shipment in order to get a seal and minimize the chances of leakage. Also, all airlines require that the seal be engaged and some will ask that seal engagement be demonstrated by the shipper prior to their accepting the body for shipment. The casket is sealed by closing the lid and inserting the casket key into the mechanism. The casket key is then turned clockwise until the lid is pulled tightly down against the bottom of the casket. The key will usually stop turning when the seal is made. Do not lose the casket key!
 Figure 21: Casket key being used to close the lid of a sealer casket.
Figure 21: Casket key being used to close the lid of a sealer casket.
 Figure 22: Sealer casket on air tray wrapped in plastic just prior to being covered with a cardboard over-container.
Figure 22: Sealer casket on air tray wrapped in plastic just prior to being covered with a cardboard over-container.
Air trays are as important to facilitating shipment of a patient in a casket as the casket itself. No airline will accept a visible casket as air cargo for public relations reasons. Since freight and baggage can usually be seen being loaded by passengers, all caskets must be covered in a way that makes them unrecognizable as such, i.e., disguises both their surface and unique shape. Airlines will reject a casket which is not covered with an air tray. Most air freight companies will stock air trays but some will not. Especially in smaller communities it is important to acquire an air tray well in advance of need or have a simple plywood box built to cover the casket. The name used by US airlines for a casket air tray is a Jim Wilson tray, an a body being shipped is referred to as a “Jim Wilson.” This is done for public relations reasons as many passengers are unaware that corpses are being transported in the cargo hold and would be distressed to learn that this is the case.
 Figure 23: Rudimentary and inexpensive in-field constructed patient air shipper.
Figure 23: Rudimentary and inexpensive in-field constructed patient air shipper.
In the event a sealer casket is not available or affordable, many mortuaries can arrange to have a plywood box built on short order, to serve as an outer box for a mortuary shipment box. Some quality mortuary shipping containers are still being manufactured, and, if one of these is available (or a poor quality shipping container can be well-caulked with silicone) it can be used as the inner box. The vertical supports used for adjustable metal shelving can be used as corner bracing and to attach the lid to the box. Trunk handles can be acquired from a hardware store to facilitate lifting, and fiberglass batting (building insulation) can be used to fill the space between the inner and outer box. Figures 23 and 24 show an air shipping containment constructed in just this fashion in the field during the early 1980′s. An exterior electrical outlet cover (for standard 110 VAC service) was used to protect a Radio Shack remote-sensing thermometer.
 Figure 24: A mortuary air shipping container positioned inside a plywood outer box and fiberglass building insulation is placed between the metal air shipper and the plywood box.
Figure 24: A mortuary air shipping container positioned inside a plywood outer box and fiberglass building insulation is placed between the metal air shipper and the plywood box.
Importance of Insulation
Some human cryopreservation organizations insist that the use of insulated air shippers is unnecessary, and cite firsthand experience with patients transported in a standard metal shipper on an air tray packed in ice during the summer months, arriving with little melting and the patient still “adequately” refrigerated. Such stories are no doubt true, and the author has been forced to ship a patient under such circumstances once in the past. However, such careless handling relies on everything going exactly as planned as well as dumb, blind luck. A patient who is accidentally loaded onto the wrong plane, a patient in an aircraft grounded for mechanical trouble or due to a bomb threat or inclement weather, can experience many hours and in some instances almost a day’s delay beyond anticipated arrival. Failure to plan for these all too commonplace contingencies is grossly negligent.
 Figure 25: Patient arrival after air shipment from central Colorado to Southern California during the summer months. The patient was a ~88 kg man whose body temperature was 20.8ºC at the time he was packed in 50 kg of ice inside a custom fabricated cryopatient air shipper insulated with 4” of EPS. Transit time was ~19 hour door to door.[3] Note that most of the ice has been converted to water during transit. The patient’s deep nasopharyngeal arrival temperature was 1.5ºC. This case points up the importance of using ample ice, especially if the patient has not yet cooled to at least 5 ºC before shipment takes place. Complex routing or other factors, such as snowstorms, hurricanes, or other severe weather that might cause the flight(s) carrying the patient to be delayed should be carefully considered. The possibility that the patient may be temporarily lost in shipment or misrouted should also be given consideration as these events can add many hours to transit time.
Figure 25: Patient arrival after air shipment from central Colorado to Southern California during the summer months. The patient was a ~88 kg man whose body temperature was 20.8ºC at the time he was packed in 50 kg of ice inside a custom fabricated cryopatient air shipper insulated with 4” of EPS. Transit time was ~19 hour door to door.[3] Note that most of the ice has been converted to water during transit. The patient’s deep nasopharyngeal arrival temperature was 1.5ºC. This case points up the importance of using ample ice, especially if the patient has not yet cooled to at least 5 ºC before shipment takes place. Complex routing or other factors, such as snowstorms, hurricanes, or other severe weather that might cause the flight(s) carrying the patient to be delayed should be carefully considered. The possibility that the patient may be temporarily lost in shipment or misrouted should also be given consideration as these events can add many hours to transit time.
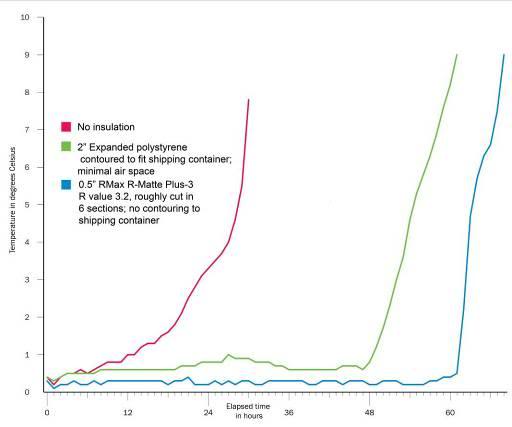 Figure 26: Temperature/time curves measured inside the transport container. Temperature was measured adjacent to the left side of the head of a phantom patient made of thermal neutral expanded polystyrene (EPS) inside a metal air shipping container loaded with 43 kg of water ice. The phantom patient was enclosed in one 3-mil lightweight body bag plus and placed inside another 20-mil heavyweight body bag. Ambient temperature was maintained between 25.5 oC and 24 oC. Data collection for each experiment were synchronized to begin when the first reading on the phantom was between 0 and 0.5oC (earlier values discarded). The air shipper was evaluated un-insulated ___, insulated with 2” of EPS ___, or insulated with ½” of RMax R-MattePlus-3(R value = 3.2) foil backed polyurethane foam insulation ___. Reproduced from: “Ice Melting Time in a Patient Shipment Container by Charles Platt, Aschwin de Wolf, and Jay Wasserlauf; Suspended Animation, Inc, April 2005.”[4]
Figure 26: Temperature/time curves measured inside the transport container. Temperature was measured adjacent to the left side of the head of a phantom patient made of thermal neutral expanded polystyrene (EPS) inside a metal air shipping container loaded with 43 kg of water ice. The phantom patient was enclosed in one 3-mil lightweight body bag plus and placed inside another 20-mil heavyweight body bag. Ambient temperature was maintained between 25.5 oC and 24 oC. Data collection for each experiment were synchronized to begin when the first reading on the phantom was between 0 and 0.5oC (earlier values discarded). The air shipper was evaluated un-insulated ___, insulated with 2” of EPS ___, or insulated with ½” of RMax R-MattePlus-3(R value = 3.2) foil backed polyurethane foam insulation ___. Reproduced from: “Ice Melting Time in a Patient Shipment Container by Charles Platt, Aschwin de Wolf, and Jay Wasserlauf; Suspended Animation, Inc, April 2005.”[4]
There have already been three cases in the less than 100 patients air shipped by all human cryopreservation organizations so far where a patient’s care was compromised (delays and/or rewarming) due to an inadequately insulated and inappropriately labeled air shipment container. These statistics should serve as ample warning that casual packaging of patients for common carrier transport is unacceptable. Figure 15-23 shows the measured rewarming rates of insulated containers versus an un-insulated container under very conservative conditions with respect to ambient temperature.[4] As can be clearly seen, insulation is absolutely essential for safe air cargo shipment of cryopatients.
Arranging For Air Cargo Transportation
Regulatory Concerns
Removal of human remains from the place of death is regulated to varying degrees in all states and most countries. The complexity and character of these regulations varies enormously, not only from state to state, but from county to county and, often, from day to night in the same location depending upon who is on duty when the call comes in. In most cases, movement of remains after legal death is governed by the following regulatory elements:
1) A signed death certificate or an agreement to sign a death certificate by the attending physician is required so the patient can be removed from the location of legal death. If the patient is a coroner’s or a medical examiners (ME’s) case, the patient will be transported to the ME’s office for autopsy or further investigation prior to release. Alternatively, a “release number” will be given by the ME or the coroner to waive autopsy or further investigation and release the patient for disposition.
2) A removal Permit or Transit Permit issued by the County Department of Health is required to transport the patient out of the county where death occurred to another county or another state. Obtaining this permit is usually contingent upon filing a completed death certificate with the Health Department which they certify as acceptable.
3) A disposition Permit or a Burial Permit is required for intermediate or long-term storage; usually within 2 to 10 days of arriving in the county, or if in the county where legal death occurred, within 2 to 10 days of death. A Disposition Permit is issued only after a certified copy of the Death Certificate and of the Removal or Transit Permit is submitted to the County Health Department where disposition or extended holding is going to occur.
While these elements seem straightforward, their implementation is anything but. Signatures required (funeral directors, physician’s, next of kin, etc.), office hours of the regulatory agencies, local mortuary or public health regulations concerning the shipment of un-embalmed bodies all must be taken into consideration and dealt with professionally and with self-assurance. The ideal person to handle this regulatory burden is a local funeral director who routinely deals with county and state bureaucrats and is thoroughly familiar with the procedures and requirements for inter-county and interstate shipment. The Transport Technician will be fully preoccupied with direct patient care and will have neither the time nor the energy to undertake arrangements for air freight transport of the patient or the associated regulatory burden.
In many jurisdictions (state or county) movement of remains by common carrier without embalming is technically not allowed. This regulation is widely ignored and is sometimes circumvented in the case of cryopatients (where there is no communicable disease posing a public health risk) by having the funeral director certify that blood washout and antibiotic treatment are the equivalent of “disinfection” of the remains. In states or counties where a requirement for embalming is strictly enforced, it may be necessary to move the patient by private carrier (a mortuary vehicle or rented van) to a neighboring jurisdiction, where such rules do not exist, or are not enforced.
The funeral director should also be given the task of making arrangements for the Transport/Standby crew, or critical elements of the crew, to accompany the patient to the cryoprotective perfusion facility. In some cases it will be desirable for key personnel to arrive sooner than it is possible to have the patient arrive. Most funeral directors are skilled at making such arrangements for families of their customers on short notice and many have a good liaison with airlines and local travel agents who can greatly facilitate reservations for staff and cargo space for the patient. Often, the ability to get any staff on any timely flight to the cryoprotective perfusion facility will be solely dependent upon the funeral director making special arrangements with the airline to book them as accompanying the human remains, typically as “family.” Many airlines will “bump” regular passengers to accommodate the next-of-kin of the deceased.
Since the 9/11 attacks on the US the requirements for being able to ship air freight have become onerous. Shippers must be vetted with the freight company and there is a requirement (often changing) for how long they must have been in business before they are acceptable shippers. Most mortuaries will qualify in this regard, and it is critical that shipping arrangements be made through the mortuary, rather than directly by the HCO, unless the HCO has approved status as an air cargo shipper. Neither Federal Express nor UPS will accept human remains or body parts for shipment.
Air Cargo Logistic Considerations
Because of time constraints to get freight loaded rapidly, air freight is often not handled with care by airport personnel. The author has observed a transport container holding a cryopatient nearly drop from a conveyor belt during loading onto an airliner, and has seen caskets containing cadavers (being shipped for conventional disposal) penetrated by forklift tines which were misaligned with the pallet under the casket, and were shoved through the side of the air tray and casket, during hasty loading (resulting in massive damage to the body within). Whenever possible, the Transport Technician should supervise the handling of the patient every step of the way, including on and off the aircraft. Due to recent terrorist acts it has become increasingly difficult for the Transport Technician to do this. Until quite recently it was usually easy for the Technician to get access to air freight facilities and the tarmac to supervise loading of the patient onto the aircraft. This is now all but impossible. However, it is still important to accompany the patient to the air freight depot and to emphasize that extra care should be used in handling the patient, and that every precaution should be taken against misrouting.
Know, in advance, the most convenient and rapidly accessible airport near the HCO’s facilities!
However, the primary consideration must be timely transport of the patient regardless of the airport used. The drive time to other airports accessible to the HCO must be weighed against earlier arrival times. In most cases only one flight will be available at the appropriate time and that will determine which airport is used. Know the alternate airports for the HCO’s location and always choose earlier arrival (and less cold-ischemic time) over convenience, or direct flights. If a direct flight and a flight with stop-overs or plane changes have almost the same arrival time, choose the direct flight, as there is less chance of misrouting of the patient.
In most cases the cooperating mortician or funeral director should be employed to handle airline arrangements since s/he will be most familiar with local shipping requirements, local carriers, and can most reliably arrange transportation to the airport and handle the Department of Health paperwork requirements.
If the weather is subfreezing, it will be critically important to emphasize to airline personnel that the patient must not be allowed to remain in unheated quarters or to be exposed to subfreezing conditions for more than an hour or two, at most.
Pulsed Vascular Rinse Transport
At this time, rigor, and the cold ischemic degradation of the blood brain barrier (BBB) and the peripheral vascular endothelium and basement membrane appear to be avoidable in cryopatients only by the expedient of continuous, asanguineous hypothermic perfusion at 2-4oC.[5-8] An alternative to continuous perfusion is the use of periodic flushes or vascular rinses using a perfusate containing the appropriate substrates.[9] Recently, a perfusate has been developed which is capable of preserving canine hearts for 24 hours in a viable condition.[10] The protocol employed for use of this perfusate involves periodic short intervals of warming the organ to allow metabolic housekeeping to take place during the flush.
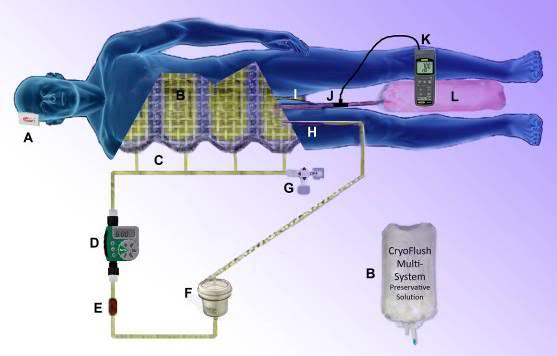 Figure 27: PulseFlush vascular rinse extracorporeal perfusion system. The system is pressurized by the body weight of the patient on a nylon mesh restrainer holding four 4 -liter bags containing the pre-chilled perfusate (B). The bags are connected via a manifold (C) to the arterial line. Pulses of flow are delivered every six hours by a modified Orbit 91213 battery operated in-line irrigation controller (D). A flow restrictor in the arterial line (E) before the auto air separating and venting arterial filter (F) establishes the maximum rate of flow from the bags into the patient. Perfusion is very low flow and pressure (~30 mmHg) into the femoral artery (H) and drainage is ‘high’ pressure (10 mm Hg) via a 22 Fr. Biomedicus flat wire femoral venous cannula (I) into a 22 liter flexible plastic effluent collection bag (L). Venous pH and temperature are logged continuously at 5 second intervals using a pH/ORP logger. Extra-cranial temperature is monitored during transport shipment using a c\temp temperature logger: http://www.temperature-data-logger.com/c_temp.html (A).
Figure 27: PulseFlush vascular rinse extracorporeal perfusion system. The system is pressurized by the body weight of the patient on a nylon mesh restrainer holding four 4 -liter bags containing the pre-chilled perfusate (B). The bags are connected via a manifold (C) to the arterial line. Pulses of flow are delivered every six hours by a modified Orbit 91213 battery operated in-line irrigation controller (D). A flow restrictor in the arterial line (E) before the auto air separating and venting arterial filter (F) establishes the maximum rate of flow from the bags into the patient. Perfusion is very low flow and pressure (~30 mmHg) into the femoral artery (H) and drainage is ‘high’ pressure (10 mm Hg) via a 22 Fr. Biomedicus flat wire femoral venous cannula (I) into a 22 liter flexible plastic effluent collection bag (L). Venous pH and temperature are logged continuously at 5 second intervals using a pH/ORP logger. Extra-cranial temperature is monitored during transport shipment using a c\temp temperature logger: http://www.temperature-data-logger.com/c_temp.html (A).
It is not now technically feasible to follow the transient warming protocol demonstrated to be most be effective in preserving cardiac (and possibly skeletal) muscle viability. However, based on experiments conducted by the author at Critical Care Research, Inc., in 1998-99, periodic vascular rinses are effective at reducing, and in some cases eliminating the onset of cold ischemic rigor in rabbits. To achieve this in human cryopatients a simple system has been developed using a modified Orbit 91213 in-line lawn irrigation controller. Four 4-liter bags of perfusate are pressurized by being placed in a custom made flexible nylon mesh holder and restrainer which the patient is placed atop in the air shipper (Figure 27). The weight of the patient pressurizes the fluid in the bags which is connected via a tubing manifold to the Orbit 91213 controller. The controller operates on two AA (1.5V) alkaline or (preferably) lithium batteries. The unit is easy to operate and program and should be set to deliver a vascular rinse pulse of 4 minutes duration (4 L) every 6 hours. Instructions for the modified Orbit controller are present as Appendix 1 to this Chapter.
The Orbit 91213 controller can be integrated into the PulseFlush tubing pack and ethylene oxide sterilized with the batteries removed (Batteries must be returned to the device prior to priming of the arterial line). The fluid contact parts of the device are biocompatible (stainless steel, polypropylene, polyethylene and Buna-N). The device performs reliably under harsh conditions (wet environment, subfreezing temperature to -8oC) and, when used in conjunction with the appropriate tubing pack (including a flow restrictor) and perfusate bags holder, it will reliably deliver 4 liters of flow at ~30-40 mm Hg pressure q. 6 hours (1 LPM/min flow rate).
If pulse vascular rinse is being used, it is important to elevate the patient on ice packs as far above the bottom of the inner container as possible (i.e., that still allows for the lid to be closed). The venous effluent collection bag should be placed on the bottom of the inner container with no ice or other material between the bag and the bottom of the inner box. This will provide the maximum amount of gravity ‘fall’ from the venous cannula to the collection bag. The venous line and the venous effluent bag must be primed and completely de-bubbled prior to activation of the system. Failure to do this will result in air-locking of the venous line and failed venous drainage.The venous drainage bag should be placed between the patent’s lower legs with no ice on top of it. Be sure to ensure that there is adequate space for the bag to fill to its 22 L volume.
Once the tubing set has been connected to the patient, the manual override on the Orbit controller should be triggered, and one 4-liter rinse given by gravity before connecting the perfusate bag manifold. This is done in order to establish that the system is functioning properly, assure that venous drainage is adequate, and prime the venous line and the effluent collection bag. Connection of the perfusate manifold must be done with minimal air introduction into the system. The arterial filter must be secured in the filter holder and positioned so that the air separation vent port is ‘upright’ and free from any obstructions. The arterial and venous lines must be carefully positioned to avoid kinking or occlusion of the lines, and be secured in place with Backhaus forceps anchored to the patient’s skin.
The patient air shipper should be clearly labeled with “This Side Up” stickers showing the proper orientation of the container.
References
1. Jones T: Suspending “Mrs. Stone (Alcor patient #1206). Cryonics 1994, 15(3):9-13.
2. Quaife A: Personal Communication re: cryopreservation of Mary DeMarr. In. Edited by Darwin M. Berkley, CA; 1973.
3. Jone T: The cryopreservation of Jim Glennie. Cryonics 1992, 13(9):10-13.
4. Platt C, de Wolf, A, Wasserlauf, J.: Ice Melting Time in a Patient Shipment Container. In.: Suspended Animation, Inc.
5. Darwin M, Leaf, JD, Hixon, H.: Case report: neuropreservation of Alcor patient A-1068. 1 of 2. Cryonics 1986, 7(2):17-32.
6. Darwin M, : Unpublished case report of Alcor Life Extension Foundation patient A-1036. In.; 1988.
7. Darwin M: Unpublished case report of Alcor Life Extension Foundation patient A-1410, . 1992.
8. Henson H: The Transport of patient A-1312. . Cryonics 1992, 13(2):18-24.
9. Fahy G: Polyglycerol compositions for the protection of living systems from states of reduced metabolism. In. Edited by Office USP. USA; 2009.
10. Fahy G, Wang, TC.: Extended Organ Presrvation. In: World Intellectual Property Organization. Edited by Organization WIP, vol. PCT/US2005/021472. USA; 2004.
Footnotes
[1] A well insulated container is one which has at least 2” of EPS or ½” of of RMax R-MattePlus-3(R value = 3.2) foil backed polyurethane foam insulation

Thanks for doing this. A real master class on this subject.
I am about to file papers to allow me to ask my goverment agency employer to transfer me to one of several locations close to Alcor: 1) southern california (LA area or Inland Empire) 2) phoenix 3) tucson 4)el paso 5) albuquerque or 6) las vegas.
We have or will have offices in all those locations by the end of the year.
you wrote:
====================
This is done for public relations reasons as many passengers are unaware that corpses are being transported in the cargo hold and would be distressed to learn that this is the case.
=============
Ah, superstition. This is why cryonics will never make any significant headway until we infuse it with elements of Christianity.
You write: “I am about to file papers to allow me to ask my goverment agency employer to transfer me to one of several locations close to Alcor: 1) southern california (LA area or Inland Empire) 2) phoenix 3) tucson 4)el paso 5) albuquerque or 6) las vegas. We have or will have offices in all those locations by the end of the year.” If I were a believer, I would say, “God help both you and Alcor.” As it is, I can only wait and watch with morbid anticipation.