By Mike Darwin

Cooling as the First Last Aid
Unarguably one of the simplest, and also the most powerful and effective cryonics first aid measures, is to cool the patient. At first glance, this would seem to require little in the way of preparation. After all, how hard is it to get ice and put it on the patient? The answer depends on the answers to two other questions: “How quickly do you want it done” and “where do you live?”
In cases of unexpected cardiac arrest, much valuable time can be lost running out to get ice. In many cases, the next-of-kin or the person on the scene cannot do this, because they must wait for the hospice nurse, the Coroner/Medical Examiner (C/ME), emergency medical technicians (EMTs), paramedics, or the patient’s physician to arrive and pronounce medico-legal death and/or release the patient from the C/ME system. There is also the obvious consideration that most people don’t have 30 pounds of ice in their home freezers, let alone 300 pounds. Even if you live down the street from a 24-hour convenience store well stocked with ice, it will take at least 20 minutes to go there, purchase the ice, transport it home, and pack it effectively around the patient. (Pay attention to that word effectively; more on that shortly.)
An Alternative to Ice?
In Europe, and most of the rest of the world, there is an added problem which severely compromises fast and effective cooling, principally that ice is simply not available most of the time, and is virtually never available in quantity, without advanced notice and preparation. Even well traveled North Americans seem unable to internalize the understanding that in most of the rest of the world, ice is a rare commodity, and it is viewed neither as a luxury, nor a necessity. What’s more, this is not simply a matter of the relative wealth of nations, but rather is rooted in cultural differences. The abundance of 24-hour retailers selling ice in North America is an artifact of North American culture and tastes. Wealthy nations, with an arguably higher standard of living (unarguably, if you consider mortality and morbidity to be the ultimate yardstick), have neither water ice in quantity, nor 24-hour convenience stores. London, one of the undisputed great cities of the world, has few 24-hour shops and virtually no easy access to ice in quantity, day or night. The same is true of Moscow, Mumbai, Munich, and Paris.
Another factor to consider is that per capita, vastly fewer non-North Americans have private transportation, and in many of the big cities of the world, including London, most or even all mass transit stops at midnight, and does not resume until 0400 or 0500.
The growing number of cryonicists living in these environments needs a workable alternative to ice, and they need a realistic approach to getting help in an emergency, because another problem that is especially acute for cryonicists in Europe is that they live quite far from each other. They are also handicapped by the fact that there are no full time personnel or often even volunteers available to help. Thus, having a workable, immediately available alternative to ice is essential.
Even in the US, cryonicists living in remote areas, small towns, or who face logistical problems in getting ice in an emergency would profit from having an “instantly” available source of refrigerant that did not require a dedicated freezer, or consume all the household refrigerator’s freezer capacity. Elderly cryonicists, and others at high risk of unexpected of cardiac arrest who cannot maintain a dedicated freezer full of ice (and change it out regularly†), could also benefit greatly from an always ready ice-substitute with an indefinite shelf life.
Ice (or more properly water) has enormous heat absorbing capacity in the form of its latent heat fusion (333.55 kJ/kg)[1] and I want to make it clear that there is really nothing else that can compete with ice as a safe, practical refrigerant for inducing hypothermia in cryopatients. In other words, ice is always the first choice for use in cooling cryopatients when it is available.
When ice isn’t available, or going out to get it would delay the start of cooling, the next best thing is ammonium nitrate and water. When ammonium nitrate is mixed with water, an endothermic (heat absorbing) reaction occurs, which absorbs 26.2 kJ of heat per mole of ammonium nitrate. This happens because ammonium nitrate, like any salt dissolving in water, breaks into its constituent ions, in this case the ammonium and nitrate ions, which absorbs energy from their surroundings. The formation of new bonds between these ions and surrounding water molecules then releases that energy. However, since ammonium and nitrate ions are relatively large, the water molecules have relatively weak interactions with their diffuse charges. So, with little thermodynamic payback during this bond formation, the immediate effect of adding water to ammonium nitrate is to reduce the temperature of the mixture to about 0.5ºC (33ºF).[2]
A mole of ammonium nitrate is 80 grams, so if we compare ammonium nitrate to the heat absorbing capacity of melting ice, the comparison is surprisingly favorable, with ice absorbing 26.68 kJ of heat per 80 grams, compared to 26.2 kJ for ammonium nitrate. Of course, this analysis omits three important points, namely that 120 grams of water are required for each 80 g of ammonium nitrate, the specific heat of water (4.18 J g−1 K−1 cp for water (liquid) at 25ºC) is almost as twice as high as a 40% ammonium nitrate and water mixture (and thus is better able to absorb heat), and that the lowest point that a water-ammonium nitrate slurry reaches is 0.5ºC, as opposed to 0 degrees C for ice (and in practice the temperature is actually ~+3ºC as opposed to 1.5 to 2.0ºC for ice under real-world working conditions).[3],[4]
 Figure 1: Instant cold packs use a binary system of ammonium nitrate (NH4NO3) and water. The product is activated by squeezing (and thus rupturing) the inner bag containing the water, initiating dissolution of the NH4NO3 prills (which are porous). NH4NO3 undergoes and endothermic (heat absorbing) “reaction” as it dissolves in water. The typical chemical cold pack quickly reached a temperature of 0.5ºC, which is maintained for ~30 minutes. There is a modest increase in cooling capacity if these cold packs are chilled before use.
Figure 1: Instant cold packs use a binary system of ammonium nitrate (NH4NO3) and water. The product is activated by squeezing (and thus rupturing) the inner bag containing the water, initiating dissolution of the NH4NO3 prills (which are porous). NH4NO3 undergoes and endothermic (heat absorbing) “reaction” as it dissolves in water. The typical chemical cold pack quickly reached a temperature of 0.5ºC, which is maintained for ~30 minutes. There is a modest increase in cooling capacity if these cold packs are chilled before use.
Ammonium nitrate cooling packs are a staple in first aid kits, and are widely used in athletics for primarily the same reason they should be used in cryonics: delay in cooling, even a slight one, reduces the effectiveness of hypothermia in mitigating injury. It is better to have a more costly, slightly less efficient product immediately available, than to lose valuable time after an injury going for ice. The most widely available “instant cold pack” product is Kwik Kold,™ manufactured by the health care giant Allegiance. Kwik Kold™ is a binary product consisting of an outer plastic bag which contains small (1-2 mm) pellets of porous ammonium nitrate (prills† ) and another bag that contains water. The product is activated by squeezing and bursting the water packet, and then mixing it with the ammonium nitrate, to begin the endothermic reaction.
Kwik Kold has a number of drawbacks, the first being its high cost. Ammonium nitrate is one of the least expensive bulk chemicals in the developed world, since it is most widely used as fertilizer. A case of 50 6” x 9” Kwik Kold packs retails for ~$165.00 US, and several cases would be necessary in situations where there would be a long delay to obtaining water ice.[5] By contrast, purified ammonium nitrate purchased from scientific suppliers would cost about a third as much, $18.00 US/kg, and ammonium nitrate purchased as fertilizer would cost only $0.50 per kg. Because ammonium nitrate has been used as an expedient explosive in terrorist bombs in the US and elsewhere (ammonium nitrate was the principal explosive used by Timothy McVeigh in the Oklahoma City bombing of the Murrah Federal Building) it can typically only be purchased in agricultural form as a bulk product in 22.73 kg (50 lb) bags, and most retailers require documentation of the end-use – and often will refuse to sell it, except to known parties (farmers or agricultural concerns in their area), or to businesses with a substantial history (both credit and operational) as well as a credible use for the product.
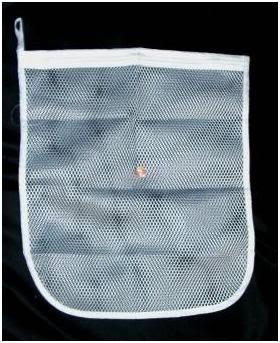 Figure 2: Coarse mesh polyester lingerie laundry bags are ideal for holding both NH4NO3 prills and water ice. They are inexpensive and widely available in a variety of sizes and shapes. The bag shown above was purchased at Walmart for less than a dollar. A penny coin is shown for scale.
Figure 2: Coarse mesh polyester lingerie laundry bags are ideal for holding both NH4NO3 prills and water ice. They are inexpensive and widely available in a variety of sizes and shapes. The bag shown above was purchased at Walmart for less than a dollar. A penny coin is shown for scale.
It is desirable to obtain ammonium nitrate in bulk not only because of of its affordability, but because it is desirable to repackage it for cryonics applications. Because Kwik Kold and similar products are dispensed in plastic bags, the refrigerating ability of the ammonium nitrate-water mixture is greatly reduced. The plastic bags limit the area of contact between the patient and the refrigerant, create micro-environments of stagnant (non-conducting) air between the bags and the patient’s skin, and slow dissolution of the ammonium nitrate in water. Ideally, the ammonium nitrate should be packaged in coarse-mesh (see Figure 2, above) bags through which water can be continuously recirculated over the patient’s head, as shown in Figure 3, below.
This simple device can be easily fabricated from off the shelf items available at most Wal-Mart (Asda or Tesco in Europe) and hardware stores, such as Home Depot, for about $90 US. This device, as well as its other applications, will be discussed in detail shortly. For now, it is sufficient to point out that the problem of purchasing bulk quantities of ammonium nitrate inexpensively needs to be solved, if for no other reason than because of the greatly increased efficiency of cooling that can be achieved. In a situation where ice may be unavailable for 12 to 24 hours, 20 to 30 kg of ammonium nitrate may be needed to hold the patient’s temperature near 0ºC. This would be cost-prohibitive for most cryonicists unless fertilizer prills are available.
Another advantage to custom packaged ammonium nitrate is that it can be stored indefinitely in air tight containers. Ammonium nitrate in instant cold pack products slowly absorbs moisture from the air, and from water vapor migrating through the plastic of the internal water reservoir bag. This results in some loss of heat absorbing capacity, but more importantly, it causes the ammonium nitrate to clump, reducing its surface area, and slowing its ability to absorb heat. Over a period of several years (depending upon storage conditions) most of the water may be lost from the water reservoir bag due to evaporation through the plastic. This would necessitate the removal of the ammonium nitrate to another bag, and the addition of a precise amount of water; hardly an acceptable option in an emergency. Nevertheless, unless and until bulk quantities of ammonium nitrate become available for last aid applications, Kwik Kold and similar products are definitely preferable to no ice at all, or to significant delays in initiating post-arrest cooling.
The message here is that there is a viable alternative to ice for non-North American cryonicists, as well those cryonicists in the US who are at high risk of unexpected death, and who cannot maintain a dedicated freezer for storage of ice (which must also be replaced frequently to prevent re-crystallization and consolidation†).
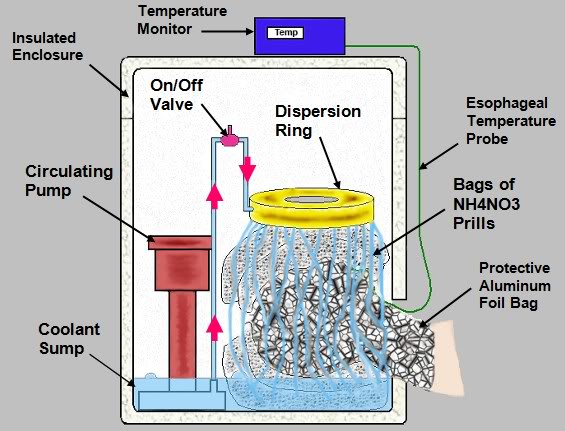
Figure 3: Schematic of a simple head refrigeration device employing nylon mesh bags filled with NH4NO3 prills through which water is recirculated. This allows for cooling with about the same efficiency as if water ice were used. See Figures 14-16, below.
The Basics of Effective External Cooling
The first requirement for maximally effective external cooling is that all of the surface area of the patient be continuously in contact with refrigerant that is as near to 0ºC as possible. It is not possible to go lower than 0ºC, because the patient would freeze in the absence of cryoprotection. This constraint on the temperature of the refrigerant has profound implications for the rate at which cooling is possible. To understand why this is so, and to understand the importance of uniform and continuous contact of the patient’s skin with the refrigerant, it is first necessary to understand how cooling occurs.
A good place to start is with the simplest situation and the one most frequently encountered in sudden and unexpected arrest, where cardiopulmonary support is not possible. This situation is simple, in the sense that the only kind of cooling that will be possible is external cooling with ice (or another appropriate refrigerant) and cooling of the head/brain will be by conduction alone. This is so because the human head is a solid consisting of gels (skin, muscle, brain) and bone. Convection does not occur in solids, and the kind of cooling we get in such a situation is called non-Newtonian cooling or non-convective cooling. Conduction cooling is comparatively “simple” because the parameters which determine its behavior are fewer than in convective cooling, where there is a complex interplay between conduction, convection and radiation†. However, while cooling within the patient’s head is purely conductive, cooling at the interface between the scalp and the other skin of the head will typically be convective, since it will involve not only conduction, but also convection (air movement in a morgue cooler, or liquid movement from melting ice, or even convection of water in an “unstirred” ice water bath).
Very recently, the French forensic pathologists Baccino, Cattaneo, Jouineau, et al.,[6] conducted a series of experiments using pig heads to empirically determine the rate of cooling under a variety of conditions, all of which are of importance to cryonicists. While pig heads are not human heads, the data from this study map the spotty and less rigorous data obtained in human cryonics cases. These data show something that may seem surprising, principally that cooling in an unstirred water bath at 0ºC is not even twice as effective as cooling in a still (unstirred) air bath at 0ºC, as shown in Figure 19 below.
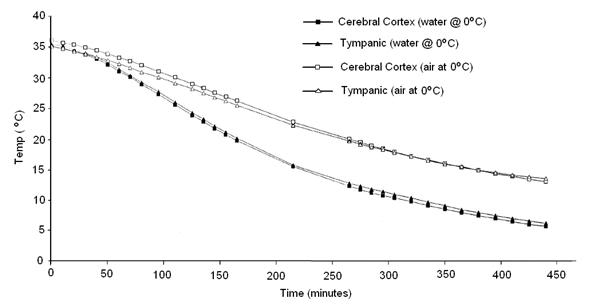
Figure 4: Cooling rate observed in porcine heads subjected to unstirred air or water cooling at 0oC from data by Baccino, et al. [6]
The reason for this is the limitation imposed by the very low value for the heat conductivity of the human head. This has the following important practical implications:
1) External conductive cooling of the human head/brain is extremely slow, even under ideal conditions of maximum surface contact with ice at 0oC, where the melt water is filmed over the patient’s head.[7],[8]
2) Once the surface of the patient’s head reaches 0oC, the brain cannot be cooled any faster regardless of the type or amount of conductive media used. In other words, using more conductive refrigerating media, or delivering them at higher flow rates than necessary to keep the skin at 0oC, will not work, and may well be counterproductive (i.e., consume limited battery power and cause splashing and aerosolization of potentially biohazardous cooling bath water).[9],[10]
3) If conditions are less than ideal because of poor contact with refrigerant (and retention of melted ice water in plastic bags) then cooling is slower still, and this is undesirable.
4) The basic requirement of uniformly cooling the surface of the patient’s head to near 0oC is, in practice, quite difficult to achieve, because holding refrigerant in contact with the patient’s head involves problems associated with melting ice, which is messy, damaging to bedding and furnishings, and can cause a slip hazard if dripped onto the floor. Containing ice (or NH4NO3 –water) in plastic bags results in considerable loss of contact with the skin, and reduces the efficiency of cooling, by causing melt water to be retained; creating a relative convective and conductive barrier. It is also virtually impossible to keep ice bags in position around the patient’s head during movement from one location to another (or for that matter, even when the patient is not being moved as can be seen in Figure 5, below)..
While there is no easy solution to problems 1 and 2 above, there is a solution to problems 3 and 4: an enclosure to hold ice or another acceptable refrigerant around the patient’s head in situations where there is no portable ice bath (PIB)†. This is a critical component of the Last Aid Kit (LAK), that should also contain a number of basic items to help improve patient care, when a Transport Team is not available. Why is having an “ice holder” to keep ice around the patient’s head so important? Again a look at Figure 5, below, is proof that a picture is worth a thousand words. The patient in this picture is being cooled with ice bags and Kwik Kold packs. As just noted, this cuts the effectiveness of ice dramatically by confining it to bags, and it is also messy, which decreases compliance and creates a real danger of slipping and falling for personnel, when tile or linoleum floors become wet and slick (something that is especially likely in institutions with well waxed and polished floors; and inside ambulances).
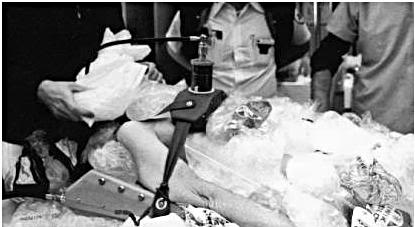
Figure 5: Cryopatient on a mechanical heart-lung resuscitator being cooled during transport using ice bags and endothermic chemical cooling packs (Kwik-Kold™).
It is also a waste of valuable personnel. The man in the vest and white shirt in the middle of the picture above was a paramedic and he was not happy about the wet floor in his ambulance. Ambulance companies in many states, and in most European countries, can be used to provide Standby and Transport assistance, as happened in the case above, but they will not be inclined to do so under conditions which expose their costly vehicles, and even more valuable personnel, to water damage.
In fact, ice in bags is such a poor refrigerant that if your head were a bowl of potato salad, you probably wouldn’t eat it if sat around at room temperature as long as the center of your head will above or at room temperature while being cooled with ice bags—there would be too much of a chance it would have spoiled. Figure 6, below, shows just how bad ice bags are at cooling, both in absolute terms, and relative to immersion in ice water (both stirred and unstirred). Even four and a half hours after the start of ice-bag cooling, the patient’s core temperature is still above room temperature: ~ 24 degrees C!
Leaving aside the grossly inefficient nature of cooling with ice bags, how well refrigerated is the patient’s head in the photo above, and how long can anyone be expected to hold those ice bags in place by hand? This photo is not unique, in fact, to the extent it is unique it is because it shows a diligent effort being made to keep this patient’s head well refrigerated. Unfortunately, many pictures of cryopatient Transports show little or no ice around the patients’ heads and, as is the case here, comparatively poor contact of the ice bags with the surface of the head.
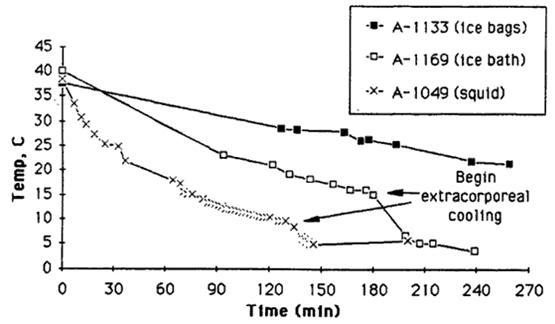 Figure 6: Comparison Of Cooling Methods: Above are actual cooling curves for three adult human cryopreservation patients receiving mechanical CPS support, using ice bags, the Portable Ice Bath (PIB), and the PIB augmented by SCCD (squid) cooling. Patient A-1133 weighed 56.8 kg, patient A-1169 weighed 57.3 kg, and patient A-1049 weighed 36.4 kg. As this data indicates PIB cooling is approximately two times as efficient as ice bag cooling. The SCCD appears to increase the rate of cooling by an additional 50%over that of the PIB (roughly adjusting for the difference in the patients’ body mass).[11]
Figure 6: Comparison Of Cooling Methods: Above are actual cooling curves for three adult human cryopreservation patients receiving mechanical CPS support, using ice bags, the Portable Ice Bath (PIB), and the PIB augmented by SCCD (squid) cooling. Patient A-1133 weighed 56.8 kg, patient A-1169 weighed 57.3 kg, and patient A-1049 weighed 36.4 kg. As this data indicates PIB cooling is approximately two times as efficient as ice bag cooling. The SCCD appears to increase the rate of cooling by an additional 50%over that of the PIB (roughly adjusting for the difference in the patients’ body mass).[11]
Increased Convenience = Increased Compliance
Some cryopatients will have minimally cooperative family, or family who are not capable of sustained cooperation due to age, infirmity, or psychological limitations. There have been cases where both husband and wife were signed-up cryonicists, and yet when one arrested the other did not pack the patient’s head in ice with a resultant 4-hour delay until brain cooling was begun.[12] Why? How could this happen? The answer is, because they were provided with neither the tools nor the instructions to respond appropriately. Resources need to be readily at hand, and members need to be repeatedly told what to do in an emergency, and given clear, easy to use instructions on how to do it. While most of you reading this may think yourselves immune to making an error such as leaving your spouses’ head unrefrigerated for ~ 4-hours, you should take into consideration the effects that advanced age, and the emotional trauma of an unexpected loss, might have on you.
Morticians do scarcely any better. What is needed is a simple device and a simple procedure which can be rapidly implemented with supplies on hand – something that the first responder only has to do only once, and from which they can then walk away from, till more skilled help arrives on the scene. If anyone questions the need for this, consider the numbers: historically ~ 30% of Alcor members have arrested with little warning, or no Standby team present. Since the Cryonics Institute does not offer Standby, presumably most of their members could benefit. Most cryonicists will not have a portable ice bath (PIB), and most will have few other resources in an emergency. So, the issue of effective cooling of just the patient’s head is non-trivial, and in practice it can only be accomplished with a device that holds the ice, or other refrigerant, in uniform and direct contact with the patient’s head.

Figure 7: A simple, expedient head ice positioner (HIP) fabricated from a plastic garment storage box and a piece of foam pipe insulation. The thermoplastic typically used to manufacture these materials is notoriously liable to fracture during cutting and drilling. The cutout in the HIP pictured above was made using a (repeatedly) heated disposable box-cutting knife
In its simplest implementation a head ice positioner (HIP) is an open-topped box with a U-shaped cut-out to accept the patient’s head as shown in Figure 7, above. This is easily and inexpensively fabricated from off-the-shelf materials and is certainly better than nothing at all. The HIP above was made from a tall plastic garment storage box (26 x 36 x 36 cm) and a section of polyethylene foam water pipe insulation, at total materials cost of $8.56 US. This design has only one serious functional impediment and that is that there is no provision for draining off ice melt before it reaches the level of the U-shaped cut-out for the patient’s neck. It would also be desirable to have a 20-liter plastic bucket or a 20 liter water carboy to collect the melt water, and allow it to be disposed of in a safe and sanitary manner (water that has been in direct contact with a patient should be treated as biohazardous waste). The drainage problem can be solved by installing a bung fitting with a threaded hose barb, or a tap near at the bottom side of the container as shown in Figure 8, below, or by installing a hand-pumped siphon device. A second overflow drain, installed just below the level of the cut-out which can be left open when the patient is not being moved (to allow water to automatically flow into the waste container) can also be incorporated into the design.

Figure 8: Off-the-shelf drain/tap kit of the kind used on ice water dispensers installed on the side of HIP to drain off melt water.
The HIP above is simple and inexpensive and will serve in situations where both personnel and ice or other refrigerant are abundant. In such situations, patient contact with the refrigerant can be facilitated by a dedicated staff or family member, excess water can be drained, and refrigerant can be replenished, as needed. However, this is far from desirable. At very least, this HIP should be equipped with an insulating shroud which reduces heat-leak into the container and conserves refrigerant. Such a shroud can be a fabric “box” with either closed cell polyethylene foam or Dacron “foam” (such as NuFoam) on all sides to provide insulation.
The importance of insulation extends beyond conserving refrigerant and eliminating the mess and hazards associated with condensation. In cases where the patient must be left unattended, such as in a C/ME’s or hospital morgue, insulation is essential to prevent rapid melting of ice by convectively stirred, and much warmer, refrigerated air. Morgue coolers vary in temperature between 4ºC and 15ºC, and typically are set in the range of 7ºC to 12 ºC. Morgue crypts in some large cities are often poorly refrigerated due to antiquated equipment and limited budgets (the latter resulting in economizing on electricity by keeping the crypt temperature high). It is thus critical to provide as much refrigerant as feasible, and to protect it against avoidable heat leak as much as possible.
Every attempt should be made to cool the core of the patient’s head to as close to 0 ºC as possible, before surrendering care of the patient to third parties, who may have little or no interest in continuing further cooling. Even under the best of circumstances, where the patient’s core (brain) temperature has reached 1-2 ºC, it must be understood that the patient’s body and neck represent a considerable heat sink and, in the case of the neck, a very considerable heat leak into the insulated enclosure of the HIP. These constraints also make it desirable that the HIP be monitored and frequently replenished with refrigerant such that the patient’s head never becomes exposed or uncovered with refrigerant.
Yet another limitation of the HIP just described is that it will not fit into some models of morgue refrigerators. While most end-loading (hatch type) morgue refrigerators have a hatch opening of 50 cm, with an available clearance of ~30 cm, most side-loading units, and some older hatch type units, have much less clearance (~20 cm).

Figure 9: Typical single body morgue refrigerator of the kind commonly used in the US, Canada and the UK.
A more sophisticated implementation of the HIP would not only hold ice or other suitable refrigerant in good contact with the entire surface of the patient’s head, but would also be insulated (excluding the neck opening) to an r-value of ≥ 3.0, with a reasonably compact insulating material that will experience minimal thermal drift over a 10-year lifespan and is non-toxic and non-irritating (acceptable materials would be closed cells foams such as polyethylene or polyester foam (NuFoam), or polyester wool batting (20 oz ). Ideally this HIP would be collapsible, and weigh no more than 5 pounds, so that even the frail elderly can handle and position it. A prototype of this device has been constructed and details will be available here in the near future.
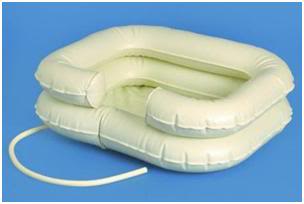
Figure 10: The EZ-Basin in-bed shampoo basin is an inflatable device designed to allow for wet shampooing of bed-fast patients’ hair. It is inexpensive ($20 US), and has the advantage of being flexible, allowing it to be used in morgue coolers and other confined spaces where a rigid HIP will not fit.
Another excellent alternative for use in morgue coolers, or other areas where space is confined, is an inflatable shampoo basin, either used as is, or modified to improve its insulating (and thus ice holding) capability (Figure 10). This secure, form-fitting basin cushions patients neck and shoulders while attendant washes hair. The EZ Shampoo basin, marketed by Medline Industries, is an inflatable basin used for shampooing the hair of people confined to bed. It is based on the same principle as inflatable plastic pools for small children, and measures 28W x 24L x 6D, and folds up for easy storage. The insulating characteristics of the basin can be greatly improved either by filling it with expanding polyurethane foam (which will set hard in a few days), or by making slits in the air cells large enough to allow it to be stuffed with Dacron polyester wool from an inexpensive pillow (Figure 11). The slits can then be closed with vinyl repair material, all temperature foil-backed gutter repair tape, or any other durable, long lasting tape that will not dry out and become brittle over time. If stuffing the EZ-Basin is being done expediently in an emergency, then duct tape may be used.

Figure 11: The EZ-Basin can be made into a more reliable HIP by filling the air cells with either polyurethane foam or Dacron polyester wool. The lack of insulation on the bottom can be addressed by placing a cut-out of closed cell polyethylene foam inside, or preferable securing a slab of the same material to the outside bottom of the EZ-Basin. Adhesive backed Velcro strips are a good way to accomplish the latter, since they allow for the foam insulation to be removed and rolled up for more compact storage.
The EZ-Basin has several disadvantages, which can be overcome with varying degrees of ingenuity and effort. The first is that there is no insulation on the bottom of the basin – just a single ~ 10 mil sheet of vinyl. This can be remedied either by attaching a sheet of 1″ or 1/2″ expanded polyethylene foam to the bottom, or placing a cut out of such foam inside the EZ-Basin, as shown in Figure 11. The second disadvantage is that it is quite shallow, with a depth of only 6″. A reasonably good compromise when using it as a HIP in a morgue cooler is to used bagged, crushed or cubed ice heaped up over the ice that is in direct contact with the patient’s skin. Finally, The EZ-Basin has no insulating top or cover. In an emergency, any reasonably good insulating material can be used to completely cover the top (and preferably the sides, as well) of the EZ-Basin, such as a blanket, fiberglass building insulation, or Dacron polyester wool insulation removed from a pillow, or purchased from a discount retailer in the form of quilter’s batting. If such open, “porous” insulation is used, once it is in place it should be covered with a sheet of plastic, such as a plastic trash bag, to improve its insulating qualities, and prevent warming of the refrigerant, and the patient’s head, due to air convection from the morgue refrigerator fan.
 Figure 12: The EZ-Basin HIP as it would be used in the event of unexpected cardiac arrest in the home on an emergent basis.
Figure 12: The EZ-Basin HIP as it would be used in the event of unexpected cardiac arrest in the home on an emergent basis.
The EZ-Basin is available from on line retailers in both the US for ~ $20:
http://www.bing.com/shopping/inflatable-ez-shampoo-basin-ez-shampoo-basin/specs/F9A27B32DB5777797B4B?q=shampoo+basin&FORM=EE
and the UK for ~ 17 pounds:
http://www.handyhealthcare.co.uk/mobility-aids/bathing/hair-washing/economy-shampoo-basin.html
Making Iceless Cooling More Efficient: A More Sophisticated Implementation
As previously noted, outside the United States and Canada, ready access to ice is problematic or nearly impossible. The problem with using chemical cooling packs in place of ice is that they rapidly lose their heat absorbing capacity and then become barriers to effective heat exchange. Additionally, since the refrigerating liquid inside the packs is not stirred and is isolated from the skin by a barrier of plastic and often trapped air as well, heat exchange is very slow and inefficient.
The solution to this problem is to construct a device which circulates the water required to drive the endothermic reaction of NH4NO3 prills with water continuously through the NH4NO3 prills and over the patient’s head. Such a device, the CephaCool, is shown in Figures 14 through 16, below.
The implementation showed below uses an inexpensive evaporative (swamp) cooler pump that operates on wall current, and is available at most hardware and large retail outlets for about $15.00 US. However, a better approach would be to use a self contained, cordless, battery operated sump or bilge pump, such as the Attwood Waterbuster Cordless Pump shown in Figure 13, below.
 Figure 13: The Atwood cordless bilge pump is a fully submersible pump that runs up to 5 hours on three alkaline D batteries (6-3/8” high x 5-1/4” diameter).and pumps up to 200 gallons per hour at a head of 4’.
Figure 13: The Atwood cordless bilge pump is a fully submersible pump that runs up to 5 hours on three alkaline D batteries (6-3/8” high x 5-1/4” diameter).and pumps up to 200 gallons per hour at a head of 4’.
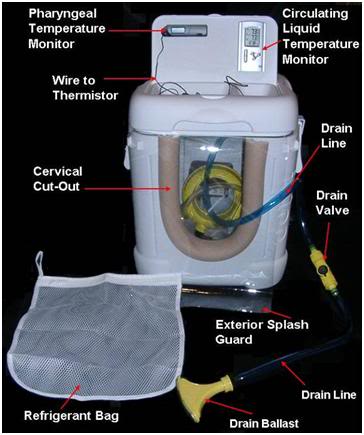 Figure 14: The CephaCool head cooling device is completely made from off the shelf items easily procurable at most hardware and large national retailers such as Target or Wal-Mart ( Asda or Tesco in the UK). The HIP portion of the device is fabricated from an Igloo beverage cooler. The refrigerant bags which hold the NH4NO3 prills are laundry bags used to contain and protect and delicate items of apparel during machine washing at home and available from Wal-Mart or Target. The drain valve, drain ballast and all connecting tubing are lawn watering items and again are available at most hardware stores as well as Wal-Mart and Target. The cushioning foam on the cervical cut-out is standard polyethylene household water pipe insulation of the kind used to reduce the risk of pipes freezing in the winter or to reduce heat loss from hot water pipes. It is available at most hardware and home improvement stores including the large national chain stores such as Lowes or Home Depot.
Figure 14: The CephaCool head cooling device is completely made from off the shelf items easily procurable at most hardware and large national retailers such as Target or Wal-Mart ( Asda or Tesco in the UK). The HIP portion of the device is fabricated from an Igloo beverage cooler. The refrigerant bags which hold the NH4NO3 prills are laundry bags used to contain and protect and delicate items of apparel during machine washing at home and available from Wal-Mart or Target. The drain valve, drain ballast and all connecting tubing are lawn watering items and again are available at most hardware stores as well as Wal-Mart and Target. The cushioning foam on the cervical cut-out is standard polyethylene household water pipe insulation of the kind used to reduce the risk of pipes freezing in the winter or to reduce heat loss from hot water pipes. It is available at most hardware and home improvement stores including the large national chain stores such as Lowes or Home Depot.
 Figure 15: The circulating pump in this implementation, as previously noted, is a low capacity evaporative cooler water pump. The diffuser ring and the on-off valve controlling flow to the diffuser ring are both lawn watering items readily available at most hardware stores and at Wal-Mart and Target. The interior splash guard was made from vinyl heet goods purchased at Wal-Mart.
Figure 15: The circulating pump in this implementation, as previously noted, is a low capacity evaporative cooler water pump. The diffuser ring and the on-off valve controlling flow to the diffuser ring are both lawn watering items readily available at most hardware stores and at Wal-Mart and Target. The interior splash guard was made from vinyl heet goods purchased at Wal-Mart.

Figure 16: The temperature monitors, both the circulating liquid temperature and pharyngeal temperature monitors are off-the-shelf consumer indoor outdoor thermometers. The pharyngeal temperature monitor shown above also has adjustable high and low alarm features. These items typically sell for $12.00 to $15.00 US and are available at hardware, auto supply, home improvement and large national chain discount retailers.
The CephaCool, as shown above, can be assembled from parts totally ~$75.00 US. With the exception of the cervical cut-out, no tools are required to assemble the components into a fully functional device. All items either slip together or are held together with industrial strength Velcro. If the Atwood cordless bilge pump is used in place of an AC evaporative cooler pump (highly recommended for safety reasons and mandatory if NH4NO3 is to be used as the coolant) the cost increases by ~$25.00 to 30.00 (the Atwood retails for ~42.00).
The CephaCool or a device like it, along with a generous supply of NH4NO3 prills should be something that every non-North American cryonics group has as a standard piece of equipment available to members upon request or for a modest fee. Members living outside of North America who are at high risk or who have relatives who are at high risk may want to consider acquiring such a device and keeping at the ready for unexpected emergencies.
How Much NH4NO3 ?
Preliminary experiments indicate that approximately 20-25 kg of ºC are required to cool a 5 kg mass of ground beef to ~4ºC. Another 5 kg is required to cool to 2ºC. Maintenance of temperature at 2-4ºC requires ~0.5 kg per hour depending upon whether or not the circulating pump is run continuously or intermittently. If operated intermittently (2-3 minutes per 30 minutes) the requirement for NH4NO3 prills drops dramatically to ~200 g/hr. It is important to understand that NH4NO3 is not a good refrigerant for maintenance cooling; there is no substitute for ice. However, it can serve as an effective “bridge” cooling material, until ice becomes available.
Cool It!
Of course, none of these tool and techniques is of any use unless they are acquired and made ready. Properly, this should be something advocated and assisted by the cryonics organizations. However, after almost 6 years of urging such actions be taken, the author has given up, and decided that the only course of action is to attempt to communicate the resources and the importance of using them directly to other cryonicists, rather than relying on any intermediary.
References
1. Mohr P, Taylor, BN, Newell, DB.: CODATA recommended values of the fundamental physical constants: 2006. Rev Mod Phys 2008, 80:633-730.
2. Bowen N: Properties of ammonium nitrate, III. J Phys Chem 1926, 30(6):736-773.
3. CRC: Handbook of Physics and Chemistry, 44th Edition; 1962.
4. Bothe J, Beyer, KD.: Experimental determination of the NH4NO3/(NH4)2SO4/H2O phase diagram. J Phys Chem A 2007, 111(48):12106-12117.
5. Cardinal: Kwik-Kold: http://www.cardinal.com/us/en/distributedproducts/ASP/103B.asp?cat=med_surg. 2011.
6. Baccino E, Cattaneo, C, Jouineau, C, Poudoulec, J, Martrille, L.: Cooling rates of the ear and brain in pig heads submerged in water implications for postmortem interval estimation of cadavers found in still water. Am J Forensic Med Pathol 2007, 28(1):80-85.
7. Gulyás B, Dobai, J Jr, Szilágyi, G, Csécsei, G, Székely, G Jr.: Continuous monitoring of post mortem temperature changes in the human brain. Neurochem Res 2006, 31(2):157-166.
8. Brinkmann B, Henssge, C, Schmitt, M, Eilers, U, Wischhusen, F. : Determination of the time of death by measurement of rectal temperature in cadavers immersed in water. Beitr Gerichtl Med 1984, 42::103-106.
9. Nelson D, Nunneley, SA.: Brain temperature and limits on transcranial cooling in humans: quantitative modeling results. Eur J Appl Physiol Occup Physiol 1998, 78(4):353-359.
10. Henssge C, Beckmann ER, Wischhusen, F, Brinkmann, B.: Determination of the time of death by measurement of central brain temperature. Z Rechtsmed 1984, 93(1):1-22.
11. Darwin M: Transport Protocol for Cryonic suspension of Humans: http://www.alcor.org/Library/html/1990manual.html. Fullerton, CA; 1986.
12. Best B: The Cryonics Institute’s 82nd Patient : http://www.cryonics.org/reports/CI82.html. 2007.
†Ice recrystalizes and consolidates into a more or less solid mass over time which it makes it very difficult to use effectively for cooling and requires expenditure of a great deal of time breaking it up into useable chunks. This process is not only time consuming but is messy and can can result injury due to haste in an emergency. Ice stored in “frost free” freezers fairly rapidly disappears due to partial melting and vaporization during the heating cycles which occur at ~6-hour intervals. These heating cycles (which vaporize ice that has formed on the refrigerating coils) also very rapidly (days) convert crushed and cubed ice into a sold mass of ice.
Footnotes
† Prills are small beads of chemicals or metals made by spraying the material into an environment (spray tower) cold enough to facilitate solidification.
† Even when ice is stored in non-frost-free chest type freezers re-crystallization and consolidation of cubed or shaved ice occurs over a period of several months.
† The first person in cryonics to consider this problem was the mathematician and cryonicist Art Quaife, who did extensive mathematical modeling of this problem in the late 1970s.
† As will be discussed at length later, a head ice positioner (HIP) should be used even when a PIB is available because it solves the problem for how to uniformly circulate cold water over the patient’s head.
